Providing better, faster treatments, adapting to the characteristics of each patient… the medical and pharmaceutical industries face major challenges to enable populations to live longer in good health. Mechanical engineering and Cetim play a decisive role in the constant quest for excellence.
Enabling humans to live longer in good health. Such is today’s — and tomorrow’s — biggest challenge for medical and pharmaceutical stakeholders. And this challenge is huge: striving continuously for better, faster results, while ensuring patient safety. These stakes concern the entire industry. Pharmaceutical companies need to shorten development times and accelerate production launches. Laboratories must go from design to production as fast as possible, while avoiding risks.
The Mobility and Healthcare Market Manager at Cetim, Vincent Caulet, sees “the pharmaceutical plant of tomorrow featuring automation, traceability and safety, especially in terms of contamination prevention, often operating at high rates of production.”
What solutions are available?
More simulations and digital twins upstream, more digitalisation and connectivity downstream, combined with technologies that guarantee hygiene and safety in critical environments.
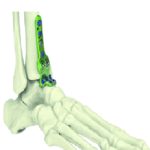
Future healthcare will ‘repair’ humans with custom-made prothesis made to last as long as possible – today, up to 30 years. It will also ‘augment’ humans, with bionic models for example. Once again, the goal is to squeeze the timeframe between the patient scan and implant procedure, through digital technologies and, especially, additive manufacturing.
Finally, the hospital of tomorrow will need exhaustive patient files, updated in real time and available to healthcare professionals at any moment, as well as equipment, namely robots, to compensate for lack of resources and to shorten in-hospital stays. “Concretely, this means a lot of digital technology, mass data management and artificial intelligence, as well as equipment maintenance management, similar to how things are run in industry,” notes Bruno Davier, Innovation Officer at Cetim.
Key know-how at Cetim
Mechanical engineers play a key role in this continuous quest. Their products are everywhere, from research labs to operating rooms, and Cetim has the valuable know-how to meet the needs of pharmaceutical stakeholders, especially concerning medical devices. From multiphysical phenomena knowledge and the related simulation tools to materials knowledge (a wide range of tests including ageing, longevity, corrosion, migration, regulated substance and material substitution), regulatory aspects, assembly techniques and robotics. Their expertise extends to air tightness, filtration and surface engineering, key elements for solving issues related to automation and preventing contamination at pharmaceutical production sites. Cetim Grand Est and Cetim Centre-Val de Loire work mainly with industry heavyweight Sanofi in this field. The Centre facility is also equipped with a laboratory with ‘cleanroom’ conditions able to test particulate cleanliness of mechanical parts of all sizes, up to 100 kg, for industry stakeholders.
Another laboratory, dedicated to biomechanics, makes Cetim a leading partner of medical implant manufacturers for the design and validation of their products. Equipped with 15 fatigue test benches and two wear simulators, each with six workstations, it carries out standardized and customized tests to demonstrate health safety for a wide range of prosthesis. Vincent Caulet notes, “Our metal additive manufacturing know-how and equipment are also available for use by this sector. And wherever companies are situated within the global healthcare ecosystem, Cetim can provide support for their transformation to help make them stakeholders in the healthcare industry of tomorrow.”
Article from Cetim Info N°259
For more information on our solutions and services dedicated to Healthcare, contact our experts: +33 (0)9 70 82 16 80 – sqr@cetim.fr